
Simulation for medical devices, as a student and a graduate | Simcenter
In 2021, I graduated from the University of Strathclyde in Glasgow, with a Master’s degree with Distinction in Biomedical Engineering. Around the world, I see many people struggle to obtain potentially life-saving treatment. The coronavirus pandemic also exacerbated existing disparities in access to healthcare. This has driven me to use my education to make the best healthcare and medical devices accessible to all.

Becoming a biomedical engineer: My university experience
During my course, I studied the design and regulation of medical devices, along with topics such as physiology and biomaterials. My main interest has always been medical device innovation. My passion then became helping to create better devices for less to promote their access. Therefore, when I saw the medical devices internship at Siemens, I applied immediately. The role has given me the chance to gain experience in simulation for medical devices and to learn more about the field.
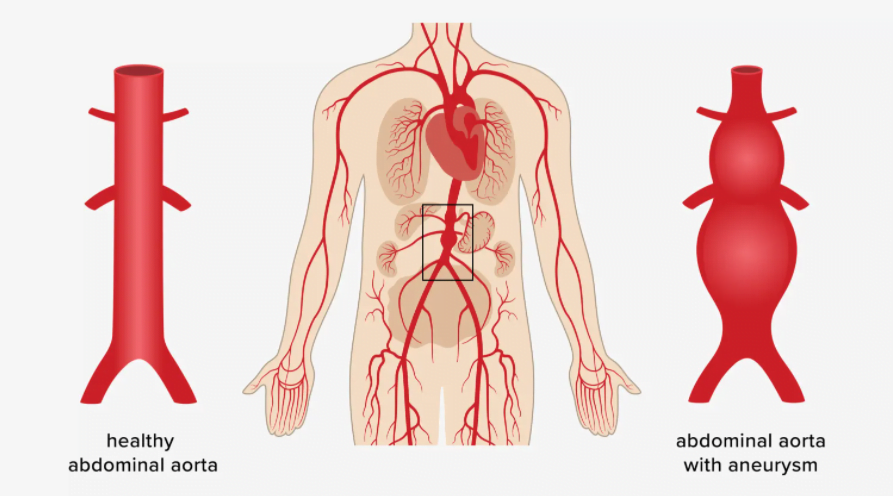
Cardiovascular diseases (CVDs) are today’s biggest global health concern. They are the leading cause of death globally1, and at least 48% of the USA population now live with some form of CVD2. CVDs can lead to the development of abdominal aortic aneurysms (AAAs). AAA prevalence is estimated to be between 4% and 8%. The risk rises with age3. Aneurysms weaken the aorta wall, causing it to bulge and, if untreated, burst. Treatment involves the replacement of the damaged vessel section with a graft. Generally, off-the-shelf grafts are used. However, it is possible to create a 3D model from patient scans to design a graft to match the patient’s anatomy. Blood flow can then be analyzed with simulation.
Simulation at university: A journey into the unknown
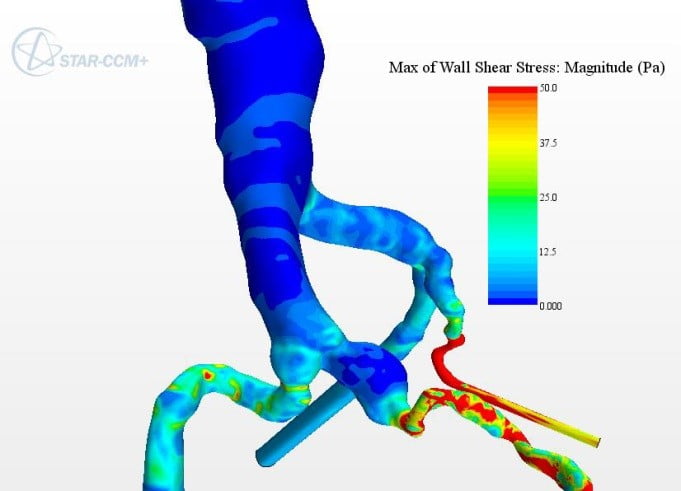
I had limited knowledge and experience of simulation for medical devices before my internship. I used it for the first time for a group project in the final year of my degree, investigating graft designs for AAA treatment. At the time I found it surprising that we were not going to use physical testing. This was because the simulation was not something I or anyone else on my course had ever used or considered at university. Of course, I was excited to be trying something new and innovative. However, I was unsure about how accurate the simulations were, assuming them to be closer to rough approximations than true digital replicas. In my mind, how could a simulation running on my computer accurately capture the complexity of human physiology?
Catching up the medical device industry
I was also unsure of the role of simulation in biomedical engineering and medical device development. I had associated it with other sectors of engineering, like aerospace and mechanical, but never my own. The medical device industry has been slow to embrace simulation, lagging behind other engineering sectors. A large part of this is due to a lack of trust in models. This is because of the risk related to medical devices. How can we be sure a model is “good enough” to be used in the development of devices upon which people’s lives may depend?
Additionally, the regulation of medical devices is extremely strict. The lack of clarity regarding simulation can make its use in device development feel risky. For many manufacturers, simulation is used only when experiments cannot be done, due to practical, ethical, or economic limitations.
Discovering the benefits of simulation for medical devices
However, once I started using simulation, I quickly began to appreciate its power. I realized how much time can be saved through removal of the reliance on physical testing. Editing a CAD model is far simpler and faster than making a new physical prototype with each design change. Also, the ability to quickly exclude weaker designs reduces wasting of time and resources. Knowing whether something is going to work on a basic level before creating a physical prototype is a huge asset to the design process. For example, early in the project it became clear that one of the graft designs would not work as it did not allow blood flow. This finding allowed us to focus our research on designs that could work clinically.
Due to the use of virtual testing, the pandemic had very little effect on my project. We were able to conduct all simulations and analysis from home on our laptops. Therefore, our work was not affected by resource delays or limited access to campus workspaces. Consequently, we were able to stay on schedule and achieve our aims. Time saved through elimination of the ineffective design enabled investigation of new areas and enhanced design exploration of each graft.
Killing coronavirus indoors with CFD simulation
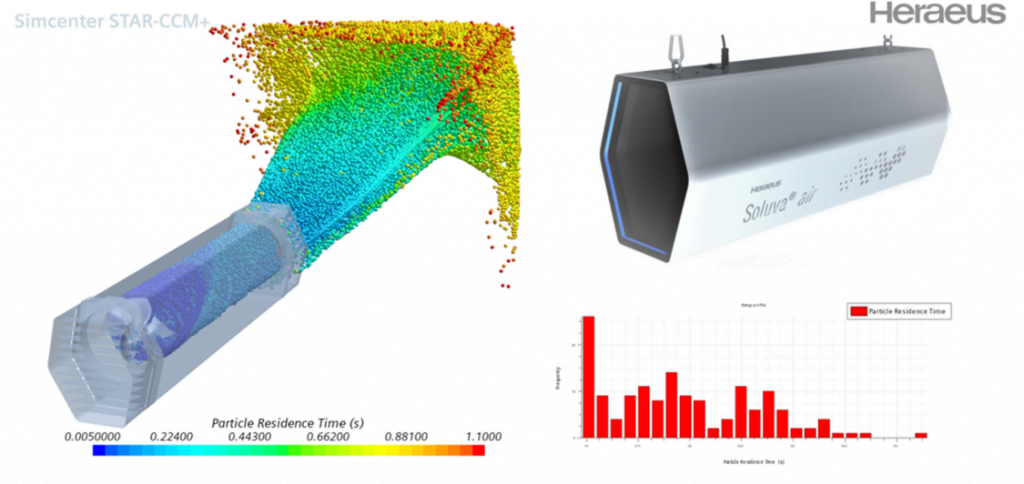
An example of this in industry is Heraeus Noblelight’s Soluva air purifier system. Their goal was to produce an air purifier that used UV-C radiation to destroy coronavirus in the air. Due to the urgency of the situation, the device had to be ready as quickly as possible. The team used simulation to optimize the design and UV-C dose. Modeling performance in the virtual space saved many production steps, and the product was ready in just 6 months. This method meant that development costs and time were reduced by ruling out weaker designs before the physical prototype stage6.
My time as a medical devices intern
My eyes now fully open to the benefits of simulation for medical devices, I began my internship in the following semester. The role provided an insight into the huge potential of simulation in medical device development. I have attended several online talks and conferences, each time learning more about what simulation can offer. Familiarizing myself with Siemens’ range of high-fidelity models I realized that simulation is more accurate than many people imagine. It can also capture all relevant physics of a system, along with the complexity associated with human body systems, both in terms of geometry and physiology.
Part of my role is applying what I learn about simulation for medical devices to promote its use in industry, highlighting the vast range of benefits and applications. I love this aspect as it helps me work towards my goal to increase the accessibility of healthcare and medical devices.
Simulation for medical devices is the key to healthcare equality
Simulation allows reduced development costs and times. This leads to lower device costs, enabling affordability for the masses. Of course, this is of huge benefit to manufacturers, but more importantly to patients. Healthcare providers are far more likely to purchase a device if it is cost-effective, along with providing a good outcome. People suffering from disease and chronic illnesses that can be remedied by a device will have equality of access to the best devices. Hence, simulation is key to closing the gap in healthcare access. I am keen to use my position to encourage its wider adoption in the industry.
As my role continues, I grow increasingly passionate about the potential of simulation for medical devices. Its ability to accelerate the innovation of medical technology while also improving patient outcomes puts it in a unique position to benefit both companies and individuals. Siemens has shown me that the use of simulation for medical devices has the power to change the industry forever.
6 months in: What I’ve gained from my internship so far
This internship has encouraged me to apply my existing skills in new ways. For example, I have used research and analytical skills gained during my studies to perform market analysis on gaps and opportunities in the medical device market. I have also improved my communication skills through regular virtual meetings with co-workers. Working remotely has meant that I have had to learn how to communicate effectively without in-person meetings.

I have also gained new skills, most crucially the ability to leave my comfort zone. When I started university, I could never have expected to end up here, working in a technology that was at that point alien to me. As I started the internship without much simulation experience, I was worried I was not ready to be jumping right into a role in something so new and innovative. However, I’ve gained so much experience in my short time here. This has given me the confidence to step out of my comfort zone in the future.
Conclusion
I never would have discovered my passion for simulation for medical devices if I hadn’t taken a leap and applied for this internship. I wouldn’t have learned as much as I have about the medical device industry’s challenges and opportunities. Therefore, I would advise other biomedical engineers to do the same as I did. If you are passionate about making a difference in healthcare for everyone, you must be bold and take a new approach. Embrace the possibilities of simulation. You’ll be making a difference not only for yourself but for the world.
Interested in finding out more about simulation for medical devices?
- Blog: Vyaire Medical – Respiratory Care in the COVID-19 Crisis
- Blog: MCAM achieves hybrid additive manufacturing strategy for new medical device mold
- On-Demand Webinar: Medical Devices: Increase innovation through multiphysics-CFD Design Exploration
References
- https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
- Heart Disease and Stroke Statistics—2019 Update: A Report from the American Heart Association, Circulation, 139(10), pp. 56–528. Accessed at: https://www.ahajournals.org/doi/full/10.1161/CIR.0000000000000659
- https://www.uptodate.com/contents/epidemiology-risk-factors-pathogenesis-and-natural-history-of-abdominal-aortic-aneurysm
- https://vascular.org/patients/vascular-conditions/abdominal-aortic-aneurysm
- https://www.medicalnewstoday.com/articles/abdominal-aortic-aneurysm
- https://new.siemens.com/global/en/company/stories/industry/2021/industrial-lighting-coronavirus-covid-19-cfd-simulation-computational-fluid-dynamics-air-purifier-uv-c-purifier-heraeus-noblelight.html

Don’t hesitate to contact Thanh for advice on automation solutions for CAD / CAM / CAE / PLM / ERP / IT systems exclusively for SMEs.
Luu Phan Thanh (Tyler) Solutions Consultant at PLM Ecosystem Mobile +84 976 099 099
Web www.plmes.io Email tyler.luu@plmes.io
